Cell Line Development
Supporting mammalian-based and microbial-based expression systems
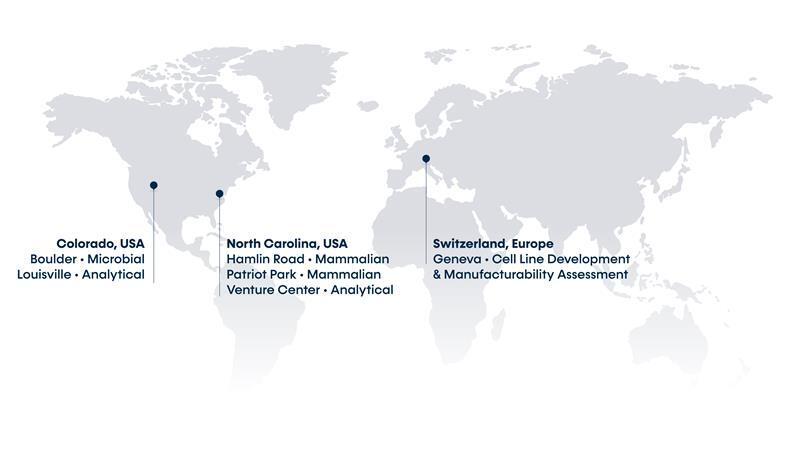
A Global CDMO Providing Innovative Biologic Solutions.
Comprehensive services for first-in-human, matched to your molecule
Developing lasting solutions to analytical challenges in biologics


Powered by Selexis® for premium, stable, and integrated CHO-based CLD.
• Versatile
• Scalable
• Efficient
A new standard of E. Coli expression for clean, efficient therapeutic protein production.
• Increased Efficiencies
• Exceptional Quality
• Engineered for Commercial Use


Learn tips & tricks on how to use the all-new Content Slider module here. Have feedback on an awesome feature we should add to this module or discover a bug? Pop right over here to submit your ideas.